#Periodic table chemistry 111 registration
This definition is for statistical and registration purposes only. Water Engineering Technology: five or more courses.Electronic Engineering Technology: five or more courses.Computer Information Systems: four or more courses.Civil Engineering Technology: five or more courses.Business Administration certificate programs: four or more courses.Business Administration diploma program: four or more courses.International Education: one ESL course.Adult Academic and Career Preparation: three or more courses or 15 hours of instruction per week.Associate of Arts or Associate of Science: nine credits.For diploma and vocational programs, all courses listed in the program outline in this calendar on a semester basis.

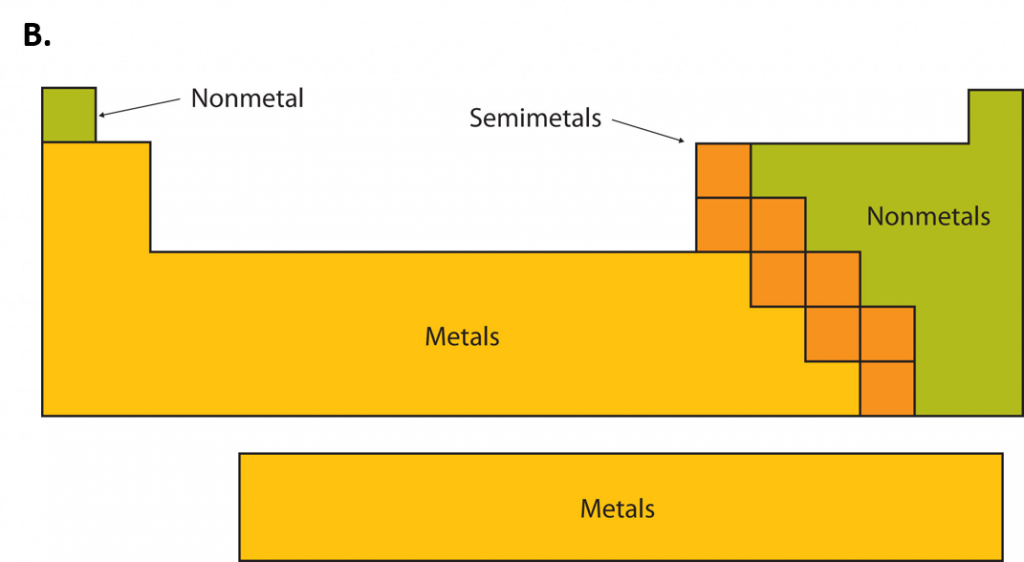
Second-year Student (associate degree and diploma programs): A student who meets the entrance requirements for a specific program has applied and been formally admitted to that program is registered in one or more of the courses which are identified as constituting the first-year or second-year requirements of that particular program and has completed 80% or more of the credits or hours towards the first-year requirements of that program.įull Course Load: For degree programs (years one to four), 15 credits per semester. A prerequisite waiver form must be signed by the department and forwarded to the Registrar's Office.Ĭredit: A credit is an assigned unit of value granted for successful completion of a course, which are used for diploma, and degree graduation requirements and/or transfer credit to another institution.Įlective: A course freely chosen from a restricted list of all Okanagan College courses, which is used to fulfill credit requirements in addition to the courses specified in the program outline.įirst-year Student (associate degree and diploma programs): A student who meets the admission requirements for a specific program has applied and been formally admitted to that program is registered in one or more courses which are identified as constituting the first-year requirements of that particular program and has completed fewer than 80% of the credits or hours toward the first-year requirements of that program. Prerequisite Waiver: Students who wish to have the course prerequisite waived, as indicated in this calendar, must receive permission from the department offering the course. Third-year Standing: Students will be granted third-year standing after completing 48 credits towards a diploma or degree program.įourth-year Standing: Students will be granted fourth-year standing after completing 78 credits towards a degree program. Second-year Standing: Second-year standing requires successful completion of a minimum of 24 credits at the 100 level or above. Courses without a prerequisite statement indicate that no prerequisite is required for enrolment. Prerequisite: A course that must be successfully completed before registration in a given course. Definitions for understanding course descriptionsĬoncurrent Registration: Compulsory registration for credit (audit registration is specifically precluded) in two or more courses at the same time even though one or more of the courses may have been previously successfully completed.Ĭorequisite: A course required to be taken at the same time as another course (audit registration is precluded) unless the course has been successfully completed before. Contact your local Okanagan College campus for up-to-date course offerings. In his honor, element 101 was named after him, mendelevium.Not all centres offer all courses listed in the calendar and courses may vary each semester. The periodic table was invented by the Russian chemist Dmitry Ivanovich Mendeleyev (1834–1907). There are also many other patterns and relationships. cesium is much more metallic than helium). For example, elements to the bottom and far left of the table are the most metallic, and elements on the top right are the least metallic. There are 3 main groups in the Periodic Table metals, metalloids, and nonmetals. The periodic table has been used by chemists to observe patterns and relationships between elements. In 1990 the International Union of Pure and Applied Chemistry (IUPAC) decided to use the new system with Arabic numerals, to replace the two old group systems that used Roman numerals. The Roman numeral names were used in most of the 20th century. There are two systems of group numbers one using Arabic numerals (1,2,3) and the other using Roman numerals (I, II, III). For example, group 18 is known as the noble gases because they are all gases and they do not combine with other atoms. Elements in a group have electrons arranged in similar ways, according to the number of valency electrons, which gives them similar chemical properties (they behave in similar ways). There are 18 groups in the standard periodic table.
The standard variation of the periodic tableĪ column of elements down the table is called a group.

